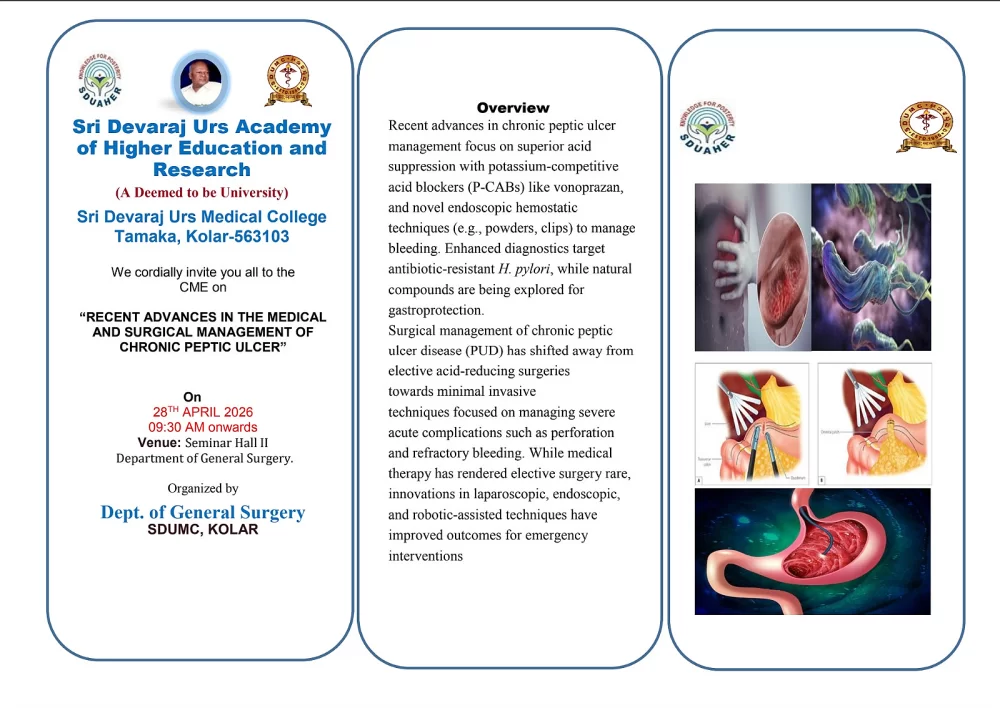
GOOD LABORATORY PRACTICE
Department of Pharmacology
CME | Year - 2025 2026 | 09 Aug, 2025
CME | Year - 2025 2026 | 09 Aug, 2025

Objectives:
1. Explain the key components of GLP regulations and OECD principles
2. Facilities, equipments, qualifications and training of personnel for the conduct of GLP studies
3. Emphasize the need for documentation, data quality, adherence to protocols, and submission of GLP reports, GLP compliance for regulatory approvals and audits 4. Explain the application of GLP principles in laboratory settings, preparation of SOPs and quality control measures in research
click here for brochure